 Disseminated magnetite has high Ti (9451940 ppm) positively correlated with Mg Al Si, and heavy Fe ( 0.76 to 1.86) and O ( 1.0 to 4. Consequently, it is more often written as 1 2 C, which is written as “ carbon-12,” ( 14 C, would thus be written as “ carbon-14.”) Nevertheless, the value of Z is commonly included in the notation for nuclear reactions because these reactions involve changes in Z. Trace element and Fe and O isotopic experiments reveal various hydrothermal precipitation environments under reduced to slightly oxidizing conditions. The subscript indicating the atomic number is actually redundant because the atomic symbol already uniquely specifies Z. The isotope of carbon that has 6 neutrons is therefore 12 6C. The symbol for a specific isotope of any element is written by placing the mass number as a superscript to the. An isotope of any element can be uniquely represented as A ZX, where X is the atomic symbol of the element, A is the mass number and Z is the atomic number. In a typical sample of carbon-containing material, 98.89% of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. The symbol for an isotope is the symbol for the element followed by the mass number. Each one has a different number of protons, electrons and neutrons. Atomic Number of Elements There are about ninety elements found on Earth. It is a hard, brittle crystalline solid with a blue-grey metallic luster, and is a tetravalent metalloid and semiconductor. The table below consists of 118 elements of the periodic table, sorted by atomic number, atomic weight, symbols, density, discovered year and the group. The table below lists the atomic numbers, symbols, and names of all the elements, with the derivations for the symbols which are not of English origin. This was shown above for the three isotopes of hydrogen. Silicon is a chemical element with the symbol Si and atomic number 14. 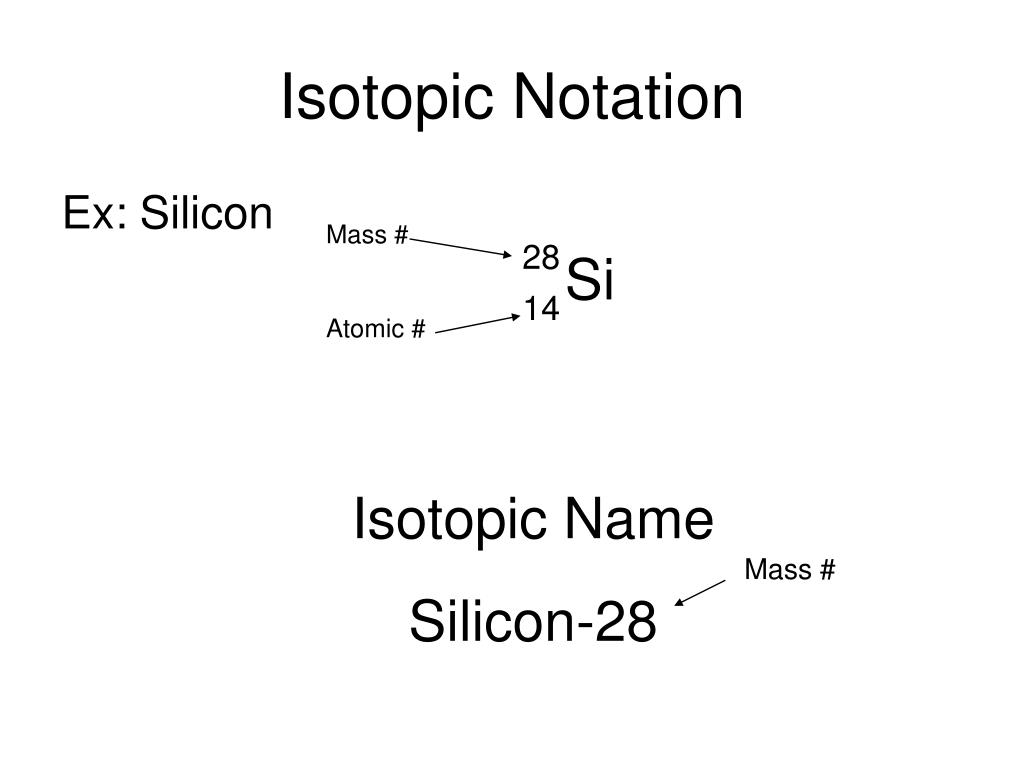
This atomic number is ordinarily given the symbol Z. An atom is first identified and labeled according to the number of protons in its nucleus. 
The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. The Parts of the Periodic Table Atomic Names and Symbols Every element has a one- or two-letter abbreviation based either on its English name or its Latin or Greek name. Every chemical element has one or more isotopes. \): Formalism used for identifying specific nuclide (any particular kind of nucleus) 
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
